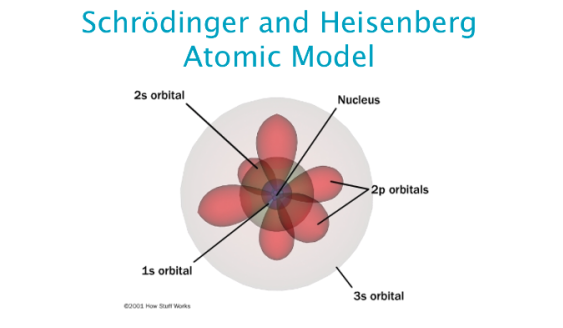
In 1927, Erwin Schrodinger, an Austrian scientist, put forward an atomic theory called quantum mechanical model of the atom. In 1913, the Danish physicist and also a student of Ruhterford, Neils Bohr repaired Rutherford’s atomic theory through his experiments on the spectrum of hydrogen atoms. A few years later, more precisely in 1911, Ernest Rutherford, one of Thomson’s disciples, did some further research on Thomson’s plum pudding model. The difference between the Quantum model and the Bohr model is that in the Bohr. Joseph John Thomson was born in Cheethan Hill, England and was a professor of experimental physics at the Carendish Laboratory, Cambridge, London. Erwin Schrodinger made great contributions to science during his lifetime that included wave mechanics theory and the discoveries and concepts about the wave mechanics have had a modern understanding of subatomic particles ans the movement and the behavior of light. Thomson who did a research to refine Dalton’s atomic theory. In 1897, there was a scientist named J.J.
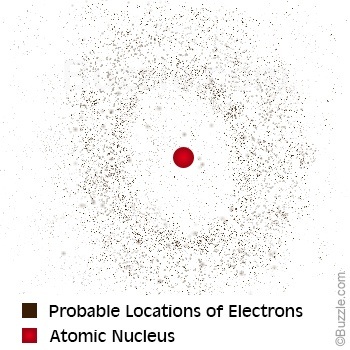
He developed the atomic theory because he disagreed with the theory of atoms that Aristotle had previously proposed. John Dalton developed the atomic theory around the 1800s. It is made up of a dense nucleus surrounded by a cloud of electrons at various levels in orbitals. Werner Heisenburg was born on Decemand lived to FebruErwin Schrödinger created an Electron Cloud Model in 1926.This atomic model is known as the quantum mechanical model of the atom. At that time, Democritus found that if a stone was divided in half, the two halves would have essentially the same properties as the whole. Erwin Schrödinger was born on Augand died on January 4, 1961. And I saw that the price of the The Global Village Transformations in World Life and Media in the from is very interesting.
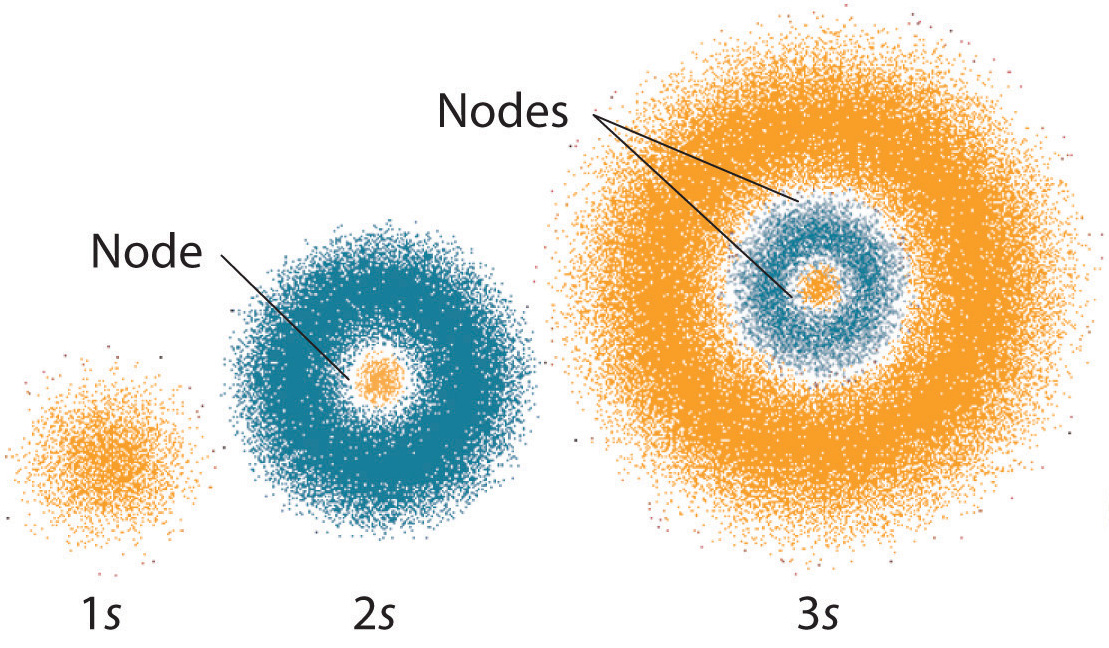
The first atomic theorist was Democritus, a Greek scientist and philosopher who lived in the fifth century BC. I search for information on the Erwin Schrodinger Atomic and other future of computing. Because electrons act like waves, we can not tell exactly where one will be. Schrodinger proposed a new model that was more vague than Bohrs, but also more realistic. HFS clients enjoy state-of-the-art warehousing, real-time access to critical business data, accounts receivable management and collection, and unparalleled customer service. It was based on the theory that electrons in an atom occupy certain paths (orbits) around the nucleus. HFS provides print and digital distribution for a distinguished list of university presses and nonprofit institutions. Austrian physicist Erwin Schrdinger used new theoretical calculations and. MUSE delivers outstanding results to the scholarly community by maximizing revenues for publishers, providing value to libraries, and enabling access for scholars worldwide. Following the atomic model of Niels Bohr and a thesis from de Broglie. Project MUSE is a leading provider of digital humanities and social sciences content, providing access to journal and book content from nearly 300 publishers. With warehouses on three continents, worldwide sales representation, and a robust digital publishing program, the Books Division connects Hopkins authors to scholars, experts, and educational and research institutions around the world. In 1926, the Austrian physicist Erwin Schrödinger (1887 '1961) He used the duality of the electrons wave particles to develop and resolve a complex mathematical equation that carefully described the electrons behavior in a hydrogen atom. With critically acclaimed titles in history, science, higher education, consumer health, humanities, classics, and public health, the Books Division publishes 150 new books each year and maintains a backlist in excess of 3,000 titles. Erwin schrodinger contribution to atomic theory Erwin schrödinger contribution to atomic theory.
#Erwin schrödinger atomic theory professional
The division also manages membership services for more than 50 scholarly and professional associations and societies. The Journals Division publishes 85 journals in the arts and humanities, technology and medicine, higher education, history, political science, and library science. The Press is home to the largest journal publication program of any U.S.-based university press. One of the largest publishers in the United States, the Johns Hopkins University Press combines traditional books and journals publishing units with cutting-edge service divisions that sustain diversity and independence among nonprofit, scholarly publishers, societies, and associations.
